Despite science’s best efforts, rare diseases are still extremely common, impacting about 300 million people around the world.
It’s time to overcome the obstacles and change the odds for patients living with rare diseases—not incrementally, but profoundly.
Breaking news. Breaking conventions.
Our goal is to do more than make headlines. It’s to completely change the narrative for patients with rare diseases.
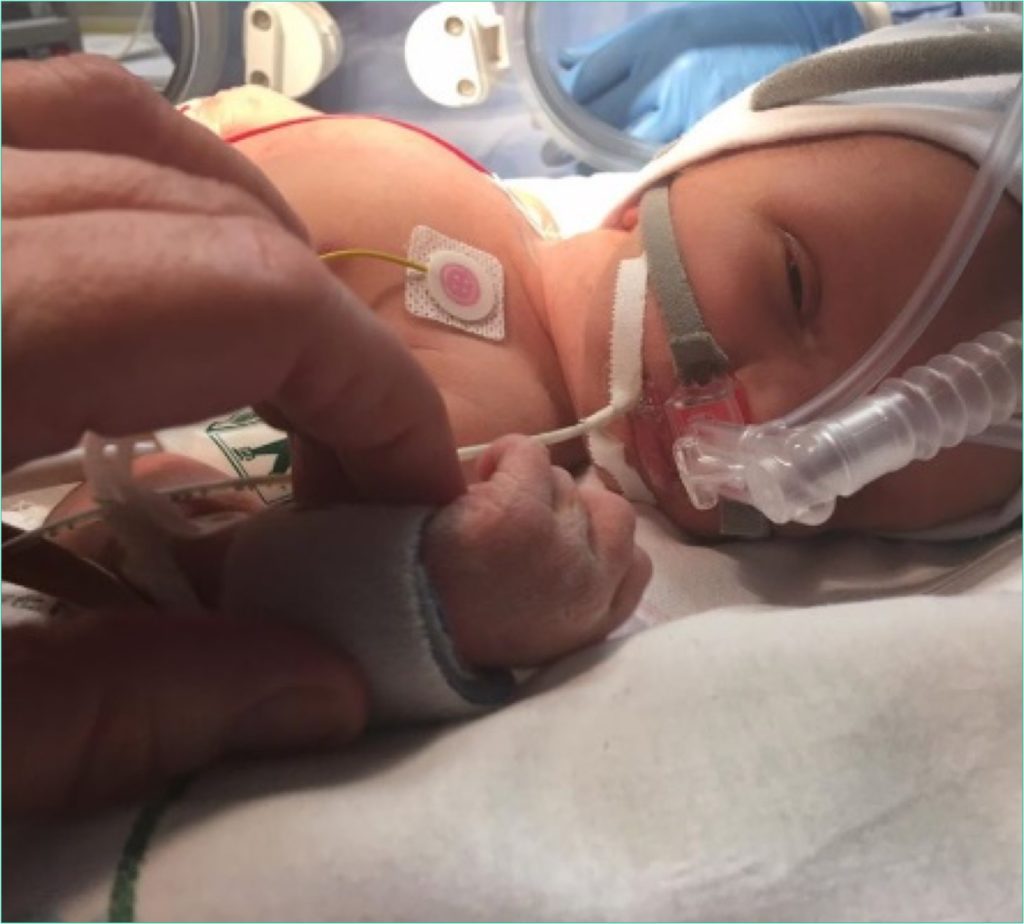

Proof that the odds are improving.
RLYB212 has achieved clinical proof-of-concept, demonstrating great promise as a safe and effective treatment option for the prevention of FNAIT.

"Our team is composed of innovators with a track record of success in translating scientific breakthroughs into life-changing treatments for people living with rare diseases. We’re starting with the development of a preventative therapy for the more than the 30,000 women whose pregnancies are at risk of Fetal and Neonatal Alloimmune Thrombocytopenia (FNAIT) each year, but we’re not stopping there."
—Steve Uden, M.D., Co-Founder and CEO
